Stem Cell Injections for Knee Arthritis
One of the primary effects of knee osteoarthritis is the degeneration of cartilage. Knee cartilage is crucial for normal joint function as it provides protective cushioning for the bony surfaces of the joint and stops them from coming into direct contact with one another. In many cases of moderate or severe osteoarthritis, the pain and disability associated with bone on bone contact is an important factor in the decision to undergo knee replacement surgery. Stem cell injections for knee arthritis may one day provide patients with a less invasive alternative to the common surgical procedures used today. Not only can stem cells potentially help relieve pain caused by knee osteoarthritis, they may also be able to slow, or even reverse the degeneration of cartilage.

What is a Stem Cell?1
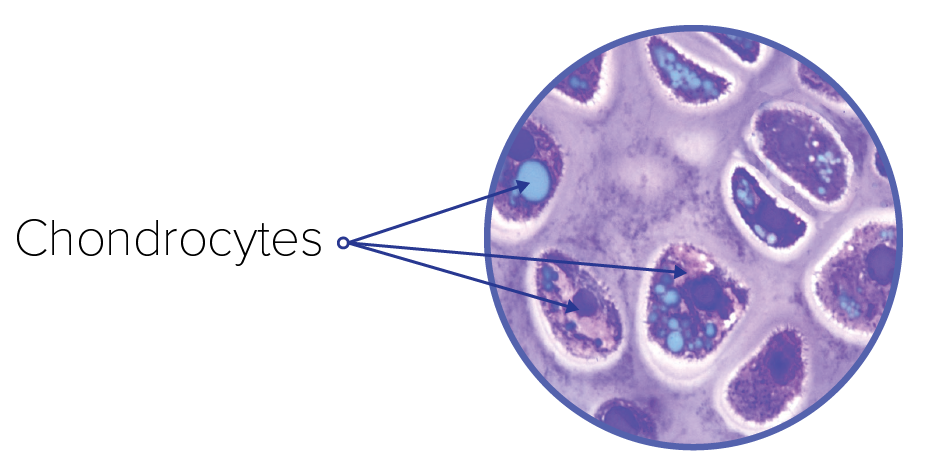
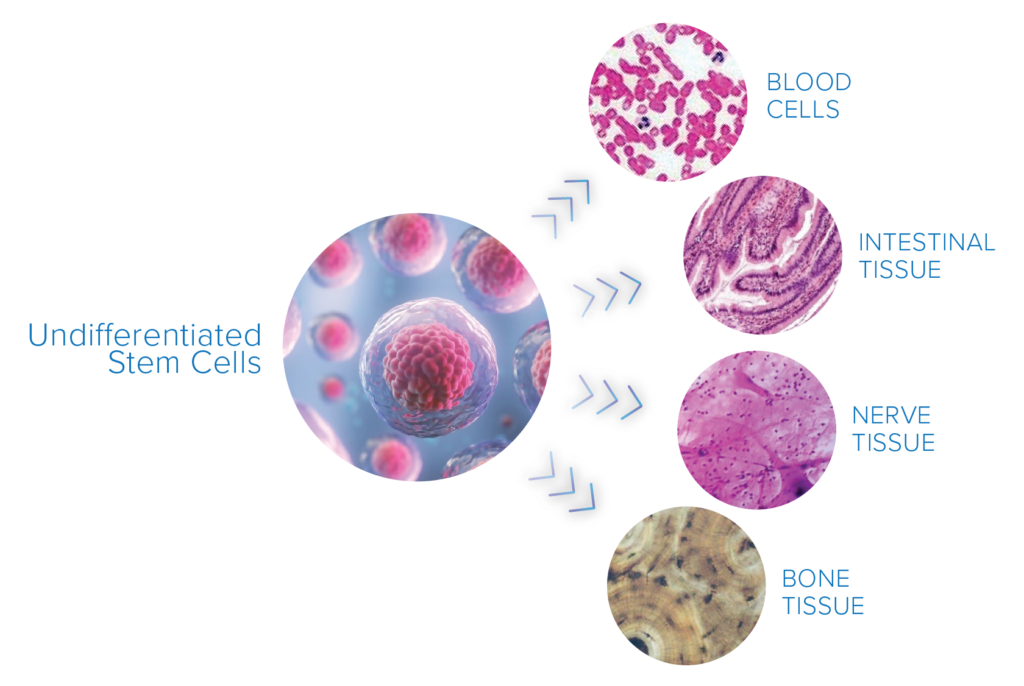
Stem cells are commonly referred to as undifferentiated or unspecialized cells. This means they have the ability to transform into more specialized cells when needed. Stem cells exist all over the body and act as a repair system for damaged organs or tissues where these specialized cell types are required. When it comes to the knee joint, it is a cell type called a chondrocyte that repairs and maintains joint cartilage. The goal of stem cell injections for knee osteoarthritis is to increase the number and activity of these cells thereby reversing or slowing cartilage degeneration.
Chondrocyte: The only cell type found in healthy cartilage. Chondrocytes produce and maintain cartilage. Reduced chondrocyte activity and volume is thought to be closely linked to osteoarthritis.2
To complicate things further, some stem cells are only capable of specializing into cell types present in their local environment. If moved to a new location, they don’t always transform into the intended cell type needed for that particular tissue. Cells with this characteristic are called Tissue Specific Stem Cells.
Stem Cell Categories
- Embryonic Stem Cell: This type of stem cell can be found in human embryos shortly after fertilization. They are unique because they can transform into any type of tissue or organ cell type in the human body. Consequently, it is possible they could be more effective than ‘adult’ stem cells which do not have this capacity. While offering significant regenerative potential, there are currently a number of ethical concerns regarding the use of embryonic stem cells.
- Tissue-Specific Stem Cell: These stem cells are found all over our bodies, and are called ‘adult’ or ‘tissue-specific’ stem cells. As mentioned earlier, these stem cells tend to be limited in their transformative capacity by being confined to change into cell types found in their local environment. Consequently, there are a large number of different subtypes of ‘adult stem cells’. For cartilage repair, the mesenchymal stem cell (MSC), a tissue-specific stem cell, is most commonly used for cartilage regeneration therapies. Mesenchymal stem cells are found in high concentrations in bone marrow and fat tissue. They have a tendency to transform into collagen and bone-related cell types which is why they are used for regenerative therapies targeted at knee cartilage.
- Induced Pluripotent Stem Cell: These cells are engineered in a lab to reliably convert into specific cell types including chondrocytes. Researchers are attempting to create versions of these that can provide effective and consistent therapeutic benefits.
Mesenchymal Stem Cells: This type of tissue-specific stem cell readily transforms into a variety of cell types including bone, cartilage, muscle and fat cells. It is the most common type of stem cell used in osteoarthritis research.3
How Do Stem Cell Injections For Knee Osteoarthritis Work?
Stem Cell Extraction
Stem cell therapy for knee osteoarthritis is most commonly done as an autologous treatment. This means that the MSCs used for treatment are extracted from the patient themselves. While MSCs can be found in many areas of the body, they are typically harvested from either fat or bone tissue. Extractions from fat tissue may yield a higher concentration of stem cells4, however, the extraction procedure (liposuction) can be more invasive than bone marrow extraction methods5. During the latter extraction procedure, a needle is usually inserted into the iliac crest area on your pelvis.
Autologous Stem Cell Therapy: This type of stem cell therapy uses your own stem cells rather than those of a donor to decrease the risk of an adverse immune reaction.
Stem Cell Culturing and Injection
Once the MSCs are extracted they are then isolated and cultured to increase their concentration and potency. This process typically takes 7-14 days, however, if the initial extraction contains a low concentration, a longer culturing time could be required6. Once the concentration of stem cells is deemed adequate they can be prepared for an intra-articular injection. This is accomplished by inserting a needle deep into the joint while being guided by ultrasound or other imaging devices7. The stem cells are then released into the damaged area of the joint.
Can Stem Cell Injections For Knee Osteoarthritis Provide Pain Relief?
The scientific research to date suggests that stem cell injections allow for a meaningful reduction in pain for those with knee osteoarthritis.8 Improvements in knee function and pain reliably exceed the required 10% threshold scientists have established for a drug to be deemed effective in this context. Some research suggests that stem cell injections may provide better pain relief than Corticosteroid injections and Non-Steroidal Anti-inflammatory Drugs8. Caution should be exercised here however, as the quality and the scale of research is not significant enough to draw firm conclusions. Further, there are many methods of harvesting, preparing and inserting stem cells; not all treatments will produce the same results making broad or generalized conclusions about the state of stem-cell research challenging.
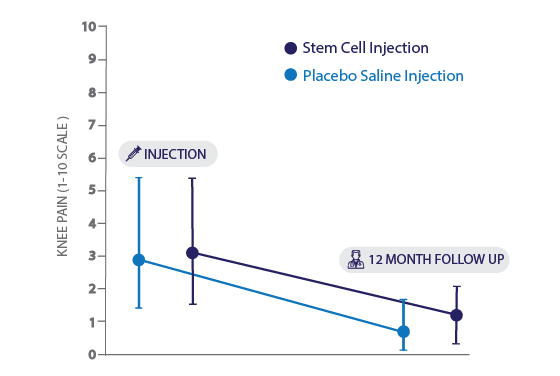
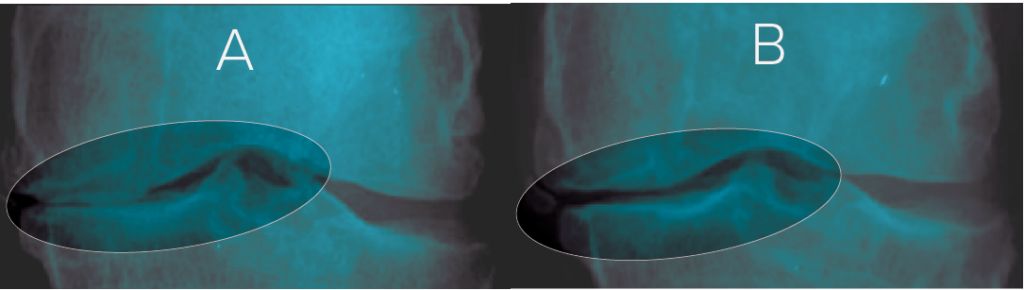
Further complicating the situation is the fact that intra-articular injections themselves are known to have a strong placebo effect. It is standard to judge any drug injection therapy against a placebo saline injection10 as this substance is low cost, fairly inert and completely safe. While there is evidence showing intra-articular stem cell injections outperform placebo injections in knee osteoarthritis patients11, there are also studies that suggest they do not. A recent study using bone marrow-derived MSCs showed that both the saline injection and stem cell injection were equally as effective at a 12 month follow-up12 (figure 1). While stem cells may offer additional benefits apart from pain relief, including cartilage regeneration, they may not exceed the performance of placebo in their ability to improve symptoms related to knee osteoarthritis. More placebo-controlled research using MSCs derived from different tissues is needed to better understand the pain-relieving effects of stem cell injections.
Stem cells may provide significant pain relief from knee osteoarthritis symptoms, however, more research is needed.
Can Stem Cell Injections for Knee Osteoarthritis Increase Cartilage Thickness?
The prospect of disease reversal is really where stem cell based therapies are exciting. Studies to date suggest that stem cell injections for knee osteoarthritis may improve cartilage quality, however, scientists have been unable to consistently demonstrate their ability to regrow cartilage in the knee13,14. One factor that may be partially responsible for the varying results in studies examining stem cell injections for knee osteoarthritis is the rehabilitation period after the initial injection(s)13.
It is well established that one of the primary causes of cartilage degeneration is joint loading15. This is why weight loss is one of the more commonly suggested strategies for slowing the progression of osteoarthritis. While a surplus of stem cells within the knee joint may have the potential to rebuild cartilage, the growth would have to exceed the rate of degeneration caused by weight-bearing activities that you perform every day.
Can better outcomes be realized by combining stem cells and joint offloading techniques?
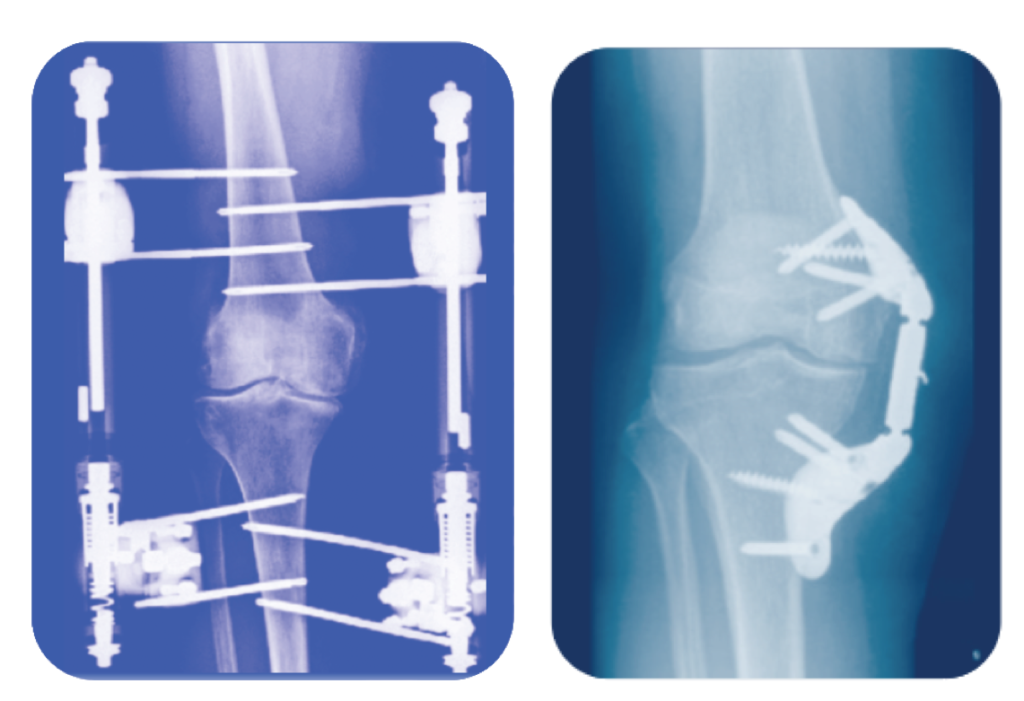
There is evidence that offloading the joint itself without stem cell therapy may increase regenerative potential. Recently, several studies showed that joint distraction techniques had remarkable benefits in cartilage regeneration15,21. This surgical procedure uses rigid external supports to gradually separate the bony ends of the joint for an extended period of time. One study21 found that after a year of treatment, cartilage thickness in participants had increased by 0.9mm. This far outpaces the average yearly cartilage loss of 0.2mm commonly seen in knee osteoarthritis. Finding like these point to a possible synergistic potential between joint offloading and stem cell therapy.
Animals studies16 examining the combination of stem cells and joint distraction have shown that these treatments when combined increased both the rate of cartilage regeneration and the quality of the tissue. While joint distraction and cartilage regeneration therapies are extremely promising, they are a surgical procedure that comes with activity limitations throughout the therapy. Further, due to the nature of having an external orthopedic device interacting internally with the joint, infections at the pin sites are common16. Because of this, they may not be suitable for all patients. Other joint offloading strategies such as weight loss, knee bracing, and mobility devices may offer alternatives to joint distraction surgery.
Alternative Offloading Devices And Stem Cell Therapy
Both wedged insoles and offloader knee braces are able to offload specific areas of the knee joint17. It is possible that this could offer a lesser, but not dissimilar, therapeutic effect as that of joint distraction combined with stem cell treatments. Recent biomechanical research has indicated that, while in a deep knee bend, Tri-compartment offloader knee braces provide a protective effect equivalent to losing 45 lbs of body weight19. This reduction in pressure may allow the therapeutic benefits of stem cells to be more effectively realized.

Many orthopedic surgeons are already using offloader knee braces in combination with stem cell therapy. The advantage of a Tri-compartment Offloader Knee Brace over surgical distraction techniques lies primarily in usability and cost. Knee braces are versatile in the sense that the offloading effect occurs immediately upon donning the brace and they can be worn during almost any weight-bearing activity.
Are Stem Cell Injections for Knee Arthritis Safe?
Stem cells can change into a variety of different cell types, therefore it can be difficult to predict exactly what will happen once they are injected. There have been a number of well-publicized cases of stem cells injected into the eye causing severe complications such as blindness. Despite this, autologous stem cell injections for knee arthritis show promise as a safe intervention, in particular when combined with joint offloading techniques. Up to 60% of patients may experience minor side effects after treatment such as swelling and knee pain13 directly after treatment. These symptoms typically gradually fade away in the weeks following the injection.
Regulatory Status of Stem Cells Injections for Knee Arthritis
While preliminary research is promising and there are numerous anecdotal success stories, the majority of regulatory bodies have not given them the stamp of approval for treating osteoarthritis.
- Health Canada updated its position on stem cell therapy in May 2019. Stem cells are classified as an ‘investigational drug’ whereby adequate clinical trials are still required to establish their efficacy and long term safety profile. This means they are not yet approved for commercial use. For patients looking to access stem cell treatments in Canada, Health Canada advises that they undergo procedures that are part of Health Canada approved clinical trials.
- The FDA published an article in the New England Journal of Medicine in 2017 detailing their perspective on stem cell therapy. The organization cautions that more research is needed to establish efficacy and safety.
Stem cells and regenerative medicine are one of the most exciting areas of research in knee osteoarthritis. If you are considering this type of treatment, remember the loading on your joint during the post-injection period influences the final outcome. Exercise, lifestyle modifications, and joint offloading devices may offer additional benefits. Consider talking to your doctor about the benefits, risks, and alternatives.
Additional Resources
National Institute of Health – Stem Cell Basics
Guide to Severe Knee Osteoarthritis
Guide to Exercise for Knee Osteoarthritis
References
- NIH Stem Cell Information Home Page. In Stem Cell Information [World Wide Web site]. Bethesda, MD: National Institutes of Health, U.S. Department of Health and Human Services, 2016
- Martin, J. A., & Buckwalter, J. A. (2001). Roles of articular cartilage aging and chondrocyte senescence in the pathogenesis of osteoarthritis.
- Uccelli, A., Moretta, L., & Pistoia, V. (2008). Mesenchymal stem cells in health and disease. Nature reviews immunology, 8(9), 726.
- Freitag, J., Bates, D., Boyd, R., Shah, K., Barnard, A., Huguenin, L., & Tenen, A. (2016). Mesenchymal stem cell therapy in the treatment of osteoarthritis: reparative pathways, safety and efficacy–a review. BMC musculoskeletal disorders, 17(1), 230.
- Pierini, M., Di Bella, C., Dozza, B., Frisoni, T., Martella, E., Bellotti, C., … & Donati, D. (2013). The posterior iliac crest outperforms the anterior iliac crest when obtaining mesenchymal stem cells from bone marrow. JBJS, 95(12), 1101-1107.
- Wagey, R., & Short, B. (2012). Isolation, Enumeration, and Expansion of Human Mesenchymal Stem Cells in Culture. Methods in Molecular Biology, 315–334. doi:10.1007/978-1-62703-128-8_20
- Bardowski, E. A., & Byrd, J. T. (2019). Ultrasound-Guided Intra-Articular Injection of the Hip: The Nashville Sound. Arthroscopy techniques, 8(4), e383-e388.
- Iijima, H., Isho, T., Kuroki, H., Takahashi, M., & Aoyama, T. (2018). Effectiveness of mesenchymal stem cells for treating patients with knee osteoarthritis: a meta-analysis toward the establishment of effective regenerative rehabilitation. NPJ Regenerative medicine, 3(1), 1-13.
- Pham, T. et al. OMERACT-OARSI initiative: osteoarthritis research society international set of responder criteria for osteoarthritis clinical trials revisited. Osteoarthr. Cartil. 12, 389–399 (2004)
- Saltzman, B. M., Leroux, T., Meyer, M. A., Basques, B. A., Chahal, J., Bach Jr, B. R., … & Cole, B. J. (2017). The therapeutic effect of intra-articular normal saline injections for knee osteoarthritis: a meta-analysis of evidence level 1 studies. The American journal of sports medicine, 45(11), 2647-2653.
- Lee, W. S., Kim, H. J., Kim, K. I., Kim, G. B., & Jin, W. (2019). Intra‐Articular Injection of Autologous Adipose Tissue‐Derived Mesenchymal Stem Cells for the Treatment of Knee Osteoarthritis: A Phase IIb, Randomized, Placebo‐Controlled Clinical Trial. Stem cells translational medicine, 8(6), 504-511.
- Shapiro, S. A., Arthurs, J. R., Heckman, M. G., Bestic, J. M., Kazmerchak, S. E., Diehl, N. N., … O’Connor, M. I. (2018). Quantitative T2 MRI Mapping and 12-Month Follow-up in a Randomized, Blinded, Placebo Controlled Trial of Bone Marrow Aspiration and Concentration for Osteoarthritis of the Knees. CARTILAGE, 194760351879614. doi:10.1177/1947603518796142
- Iijima, H., Isho, T., Kuroki, H., Takahashi, M., & Aoyama, T. (2018). Effectiveness of mesenchymal stem cells for treating patients with knee osteoarthritis: a meta-analysis toward the establishment of effective regenerative rehabilitation. NPJ Regenerative medicine, 3(1), 1-13.
- Soler, R., Orozco, L., Munar, A., Huguet, M., López, R., Vives, J., … & Garcia-Lopez, J. (2016). Final results of a phase I–II trial using ex vivo expanded autologous mesenchymal stromal cells for the treatment of osteoarthritis of the knee confirming safety and suggesting cartilage regeneration. The Knee, 23(4), 647-654.
- Messier, S. P., Beavers, D. P., Loeser, R. F., Carr, J. J., Khajanchi, S., Legault, C., … & DeVita, P. (2014). Knee-joint loading in knee osteoarthritis: influence of abdominal and thigh fat. Medicine and science in sports and exercise, 46(9), 1677.
- Goh, E. L., Lou, W. C. N., Chidambaram, S., & Ma, S. (2019). The role of joint distraction in the treatment of knee osteoarthritis: a systematic review and quantitative analysis. Orthopedic research and reviews, 11, 79.
- Harada, Y., Nakasa, T., Mahmoud, E. E., Kamei, G., Adachi, N., Deie, M., & Ochi, M. (2015). Combination therapy with intra‐articular injection of mesenchymal stem cells and articulated joint distraction for repair of a chronic osteochondral defect in the rabbit. Journal of Orthopaedic Research, 33(10), 1466-1473.
- Orishimo, K. F., Kremenic, I. J., Lee, S. J., McHugh, M. P., & Nicholas, S. J. (2013). Is valgus offloader bracing effective in normally aligned individuals: implications for post-surgical protocols following cartilage restoration procedures. Knee Surgery, Sports Traumatology, Arthroscopy, 21(12), 2661-2666.
- Budarick, A., MacKeil, B., Fitzgerald, S., & Cowper-Smith, C. (2019). Design and Mechanical Evaluation of a Novel Multi-Compartment Offloader Knee Brace. Journal of Biomechanical Engineering.
- Nebelung S, Gavenis K, Lüring C, Zhou B, Mueller-Rath R, Stoffel M, Tingart M, Rath B (2012) Simultaneous anabolic and catabolic responses of human chondrocytes seeded in collagen hydrogels to long-term continuous dynamic compression. Ann Anat 194:351–358
- Intema, F., Van Roermund, P. M., Marijnissen, A. C., Cotofana, S., Eckstein, F., Castelein, R. M., … Lafeber, F. P. (2011). Tissue structure modification in knee osteoarthritis by use of joint distraction: an open 1-year pilot study. Annals of the rheumatic diseases, 70(8), 1441–1446. doi:10.1136/ard.2010.142364
- Emrani PS, Katz JN, Kessler CL, et al. Joint space narrowing and Kellgren-Lawrence progression in knee osteoarthritis: an analytic literature synthesis. Osteoarthr Cartil 2008;16:873–82
Frequently asked Questions
Stem cell therapy for knee osteoarthritis shows promise as a treatment that may reduce pain, improve the quality of knee cartilage, and facilitate cartilage regeneration.
Stem cell injections for knee osteoarthritis are show promise as a safe intervention. The most common complications are joint swelling and soreness after the injections. However, Stem Cells are not yet approved by many regulatory bodies.
The average cost for stem cell therapy in the United States is approximately $5000.00.